A surfactant is a surface active agent known for its ability to clean dirt and create foam. Understanding which natural surfactants to choose is one of the most important formulation skills you can develop for making cleansers, shampoos and body washes.
How a Surfactant Works
Surfactants consist of two separate components — the hydrophilic (water-loving) head, and the hydrophobic (water-hating) tail. Dirt attached to skin is often oil-soluble, so when a cleansing product is applied, the surfactant lowers the surface tension of water. The hydrophobic tail attaches to dirt and oil, assembling into a geometric pattern called a micelle. The micelles — now surrounding the oil and dirt — are rinsed away with water.
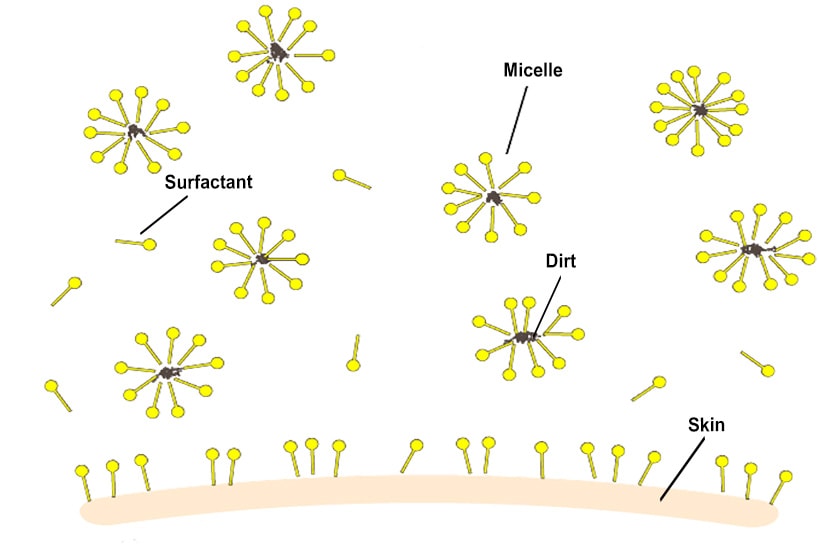
Contrary to popular belief, you do not need a lot of foam to clean properly. Foam is simply air trapped by the surfactant — we only need enough surfactant to lift oil and dirt from the surface.
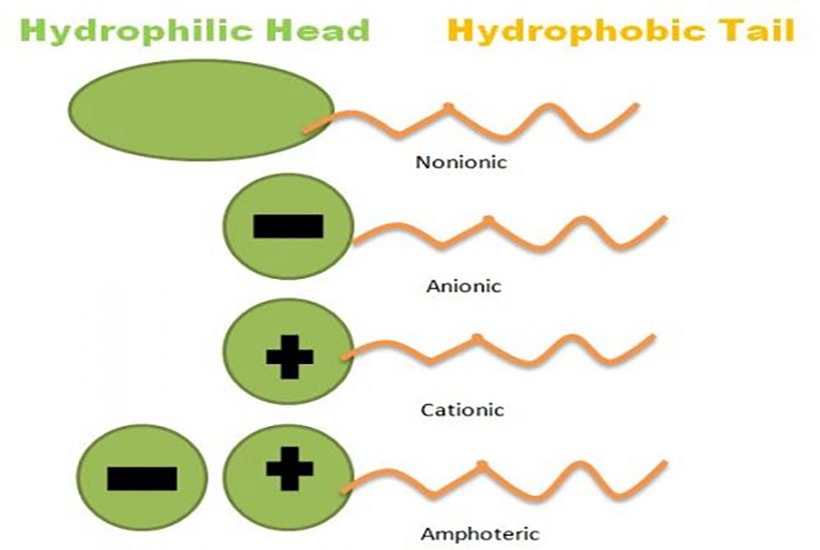
There are 4 types of surfactants based on their charge: nonionic, anionic, cationic and amphoteric.
Learn to formulate your own natural cleansers — from scratch.
My e-books teach you the science behind the ingredients — not just recipes to copy. Once you understand how formulation works, you can create cleansers, toners, serums and more, tailored exactly to your skin.
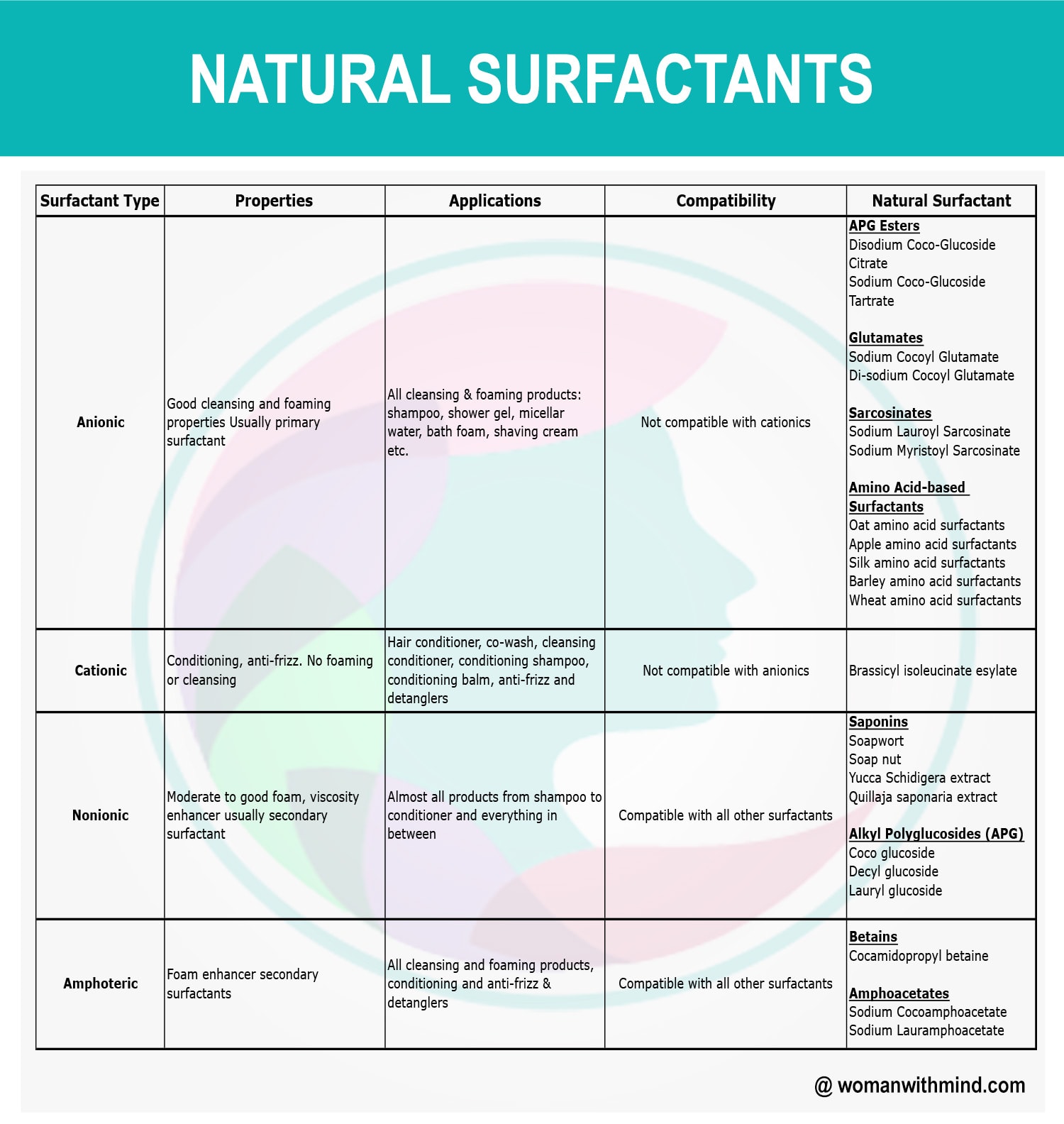
The 4 Types of Natural Surfactants
Anionic Surfactants
The most commonly used surfactants — they deliver high foaming, high cleansing and high washing capabilities. They have a negatively charged water-loving head. However, they can be harsh on skin, so it is best to combine with milder or amphoteric surfactants to balance them out.
Shampoo, shower gel, micellar water, bath foam, shaving cream, face wash
Examples: Disodium Coco-Glucoside Citrate, Sodium Coco-Glucoside Tartrate, Sodium Cocoyl Glutamate, Sodium Lauroyl Sarcosinate, oat/apple/silk/barley amino acid surfactants
Amphoteric Surfactants
Amphoteric surfactants can carry either a positive or negative charge depending on the pH of the product — making them highly adaptable. At lower pH they give nourishing, conditioning properties. At higher pH they work more like an anionic surfactant with high foaming. When coupled with anionic surfactants, they reduce harshness and stabilise foam.
All cleansing and foaming products, conditioning and anti-frizz, detanglers
Examples: Cocamidopropyl Betaine, Sodium Cocoamphoacetate, Sodium Lauramphoacetate
Cationic Surfactants
The opposite of anionic surfactants — they have a positively charged head. When used alone, they do not have high foaming capabilities. Their positive charge is attracted to the negative charge in hair, reducing electrostatic charge and making hair more manageable. They work well with amphoteric and nonionic surfactants but are not compatible with anionic surfactants.
Hair conditioner, co-wash, conditioning shampoo, conditioning balm, anti-frizz, detanglers
Natural example: Brassicyl isoleucinate esylate — the only non-quaternary cationic surfactant accepted in natural formulations.
Nonionic Surfactants
Nonionic surfactants have no charge in their water-loving heads — making them the most compatible with all other surfactant types. They are excellent as emollients, softening and soothing skin. They have no foaming capabilities and are rarely used as the main surfactant, but are invaluable as secondary surfactants to reduce irritants and improve texture. Recently, sugar-based nonionic surfactants have been developed as safer, non-toxic alternatives.
Almost all products — shampoo, conditioner, body wash and everything in between
Examples: Soapwort, Soap nut, Yucca Schidigera extract, Coco glucoside, Decyl glucoside, Lauryl glucoside
Why Formulators Blend Surfactants
Finished products rarely use a single surfactant. Blending two or more surfactants is common practice to:
- Reduce sensitisation and enhance mildness on skin
- Improve foam volume, texture and stability
- Enhance the viscosity and overall texture of the product
Guides & Printables for Skincare Formulators
These e-books and printables are designed to complement what you've learned about natural surfactants — helping you formulate complete cleansing products from scratch.

Natural Cleansers Formulation
Learn to formulate pH-balanced cleansers using the right natural surfactants — formula flowcharts and sample recipes included.

Skincare Formulation Super Bundle
Every formulation guide in one bundle — cleanser, toner, serum, balm, scrub and more. Save 20%.

Recipe Formula Calculator
Convert your formulas from percentages to weight automatically. Built-in warning alerts if ingredient ratios are out of range.

Carrier Oils & Its Properties
A 1-page reference table of common carrier oils and their skin properties — ideal for choosing the right oils for your formulas.
More Formulation Guides
Continue learning with these related guides from the Woman With Mind blog.
Conclusion
Understanding the four types of natural surfactants — anionic, amphoteric, cationic and nonionic — gives you the foundation to formulate any cleansing product from scratch. The key is knowing how to blend them to achieve the right balance of mildness, foam and texture for your specific product and skin type.